|
1/18/2024 0 Comments Ir spectroscopy absorption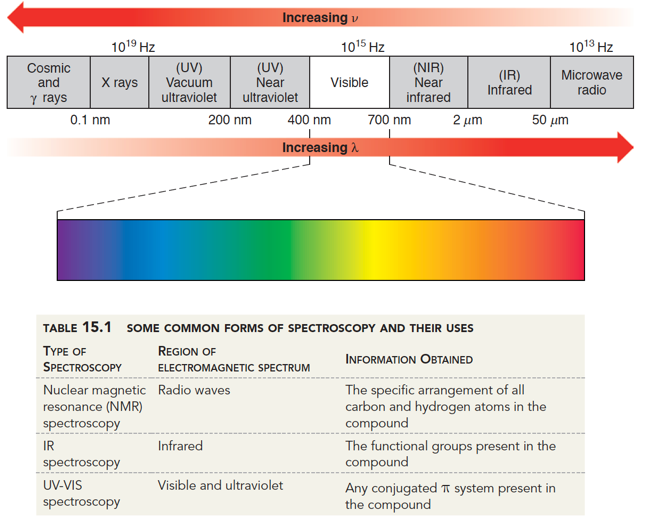
By dividing the amount of photons of a given frequency for the sample by the amount of photons from the reference, the percent absorption can be calculated. An IR SpectrumĪfter the computer module has received the data from the photodetectors for both the reference path and the sample, the computer calculates the amount of each frequency of photon along the detector. The mirror on the right is moved to vary the path length. Fourier Transform Infrared Spectrophotometer Diagram. Computerization can then produce a standard IR spectrum from the path length and interference data using a function known as a Fourier transform.įT-IR has the advantage of being much quicker for high throughput applications (such as for detection of samples from liquid chromatography (LC-IR)), but usually sacrifices some resolution compared to the traditional instrumentation. Importantly, the FT-IR instrument shifts a mirror involved in the beam splitting, altering the light’s path length, which produces a unique spectrum of IR light at each mirror position. These split beams recombine to form a unique spectrum of IR light that passes through the samples. A set of optics collimates and splits a beam almost evenly over the IR frequencies. However, the instrumentation is completely different. Fourier Transform IR Spectroscopyįourier Transform Infrared Spectroscopy (FT-IR) is an analytical technique that measures the same data as traditional infrared spectrometers. The differences in light are compared by a detector, and the results are interpreted by a computer into the familiar IR spectrum. The beams then pass through a sample and a blank reference. Infrared light is emitted from an emitter and split. The result from the detector is processed into the traditional IR spectrum. Light is released from the emitter, through the sample, then diffracted onto the detector. The Infrared Spectrophotometer Diagram of a traditional IR Spectrometer. Organic molecules absorb plenty of infrared light (but typically not much visible light because most organic compounds are white solids), making them particularly suited for analysis in the UV spectrum. It can be very useful to analyze the amount of light absorbed and reflected by objects to gain insight into the molecules and properties present. Objects that appear black absorb all visible light, likewise, objects that appear white reflect all visible light. Plants look green because they absorb red and blue light while reflecting the green back towards our eyes. 
This contributes to the colors we see (or don’t see). Upon interaction, objects absorb incident light. Human eyes are not able to see infrared light, but it exists just as much as the colors we see. However, just beyond red light is infrared light (>700nm). Humans can only see a small portion of these wavelengths, from 400 to 700 nanometers. The length of these waves determines the color we see. Light consists of individual particles, called photons, which move in a wave like pattern. Principles of IR Spectroscopy What is Infrared Light Organic Structure Elucidation Using NMR and IR.In this article, we will explore the science behind IR spec, the equipment, and some techniques for elucidating the structure of organic molecules using an infrared spectrum. Infrared spectroscopy (IR spectroscopy) is a useful analytical technique for analyzing organic molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
